We continue to seek new in-licensing opportunities that will contribute to KAKEN's sustainable growth, as well as out-licensing of the products from in-house drug discovery. We are also working to strengthen relationships with existing partner companies.
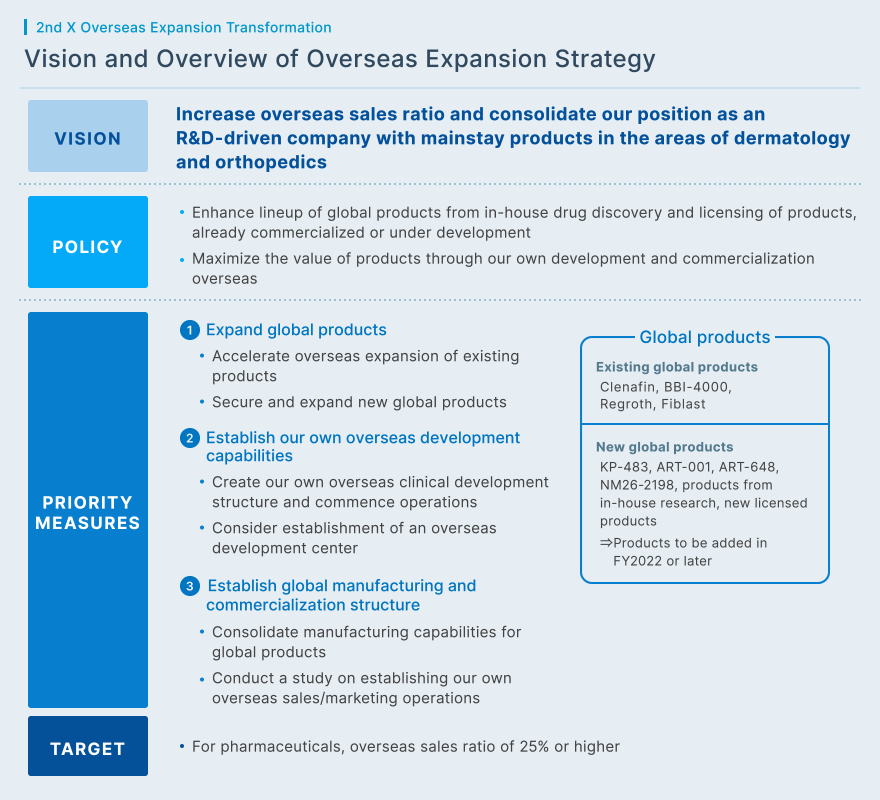
To make onychomycosis treatment Clenafin available to patients around the world, we are cooperating with partners in Europe, the United States and other countries, and are steadily expanding our collaborative network. We will continue to seek alliances with new partners, while strengthening relationships with existing licensees to maximize the value of Clenafin. Clenafin's success has significantly boosted KAKEN's name recognition and presence in dermatology. Another benefit is that an increasing number of domestic and overseas companies have approached us about potential partnerships. We will take advantage of this opportunity to pursue in-licensing and out-licensing agreements that will contribute to KAKEN's sustainable growth.
In our effort to seek new collaborative opportunities, we are expanding the scope of our drug discovery from mainly small molecules to other modalities. As we take a broader view of therapeutic areas, we will enter new areas related to dermatology and orthopedics. When we evaluate products for licensing, we determine whether they are suitable for development not only in Japan but also worldwide.
Regarding licensing, we have initiated efforts to out-license primary axillary hyperhidrosis treatment Ecclock in major Asian markets, and have been approached by a number of interested pharmaceutical companies in the region. We are now working to develop Ecclock into a next strategic product that will contribute to our overseas growth after Clenafin. Furthermore, we are focusing on overseas expansion of products from in-house drug discovery. These product include Fiblast, a wound-healing agent, and Regroth Dental Kit, a medicinal product for periodontal regeneration.
Long-Term Business Plan 2031